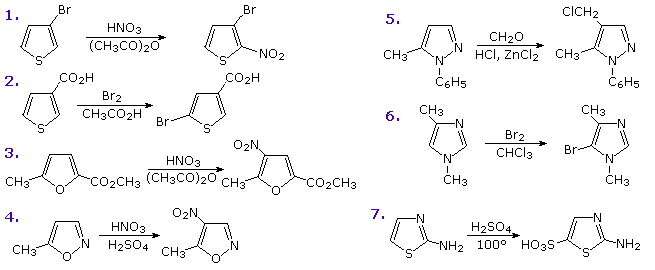
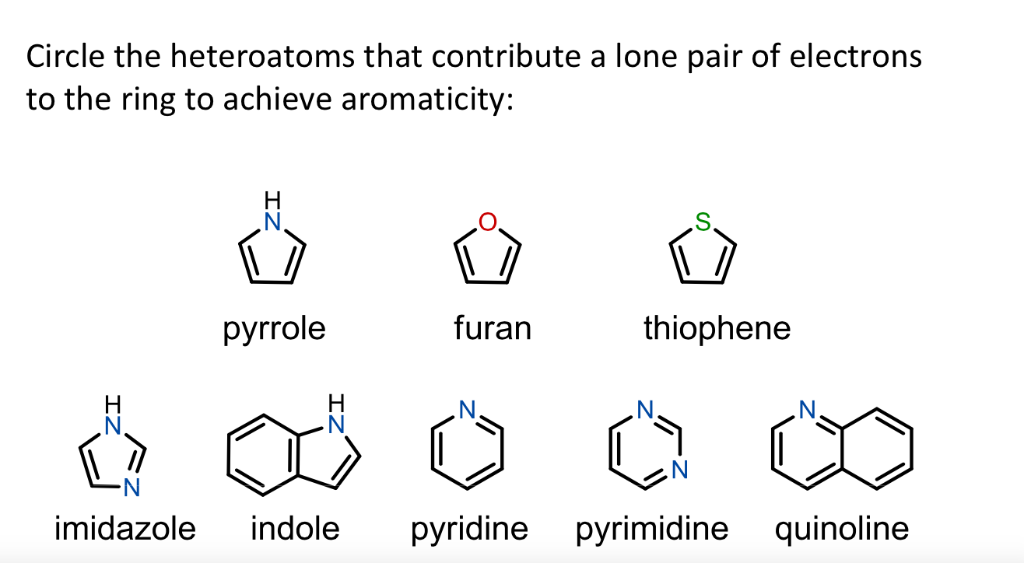
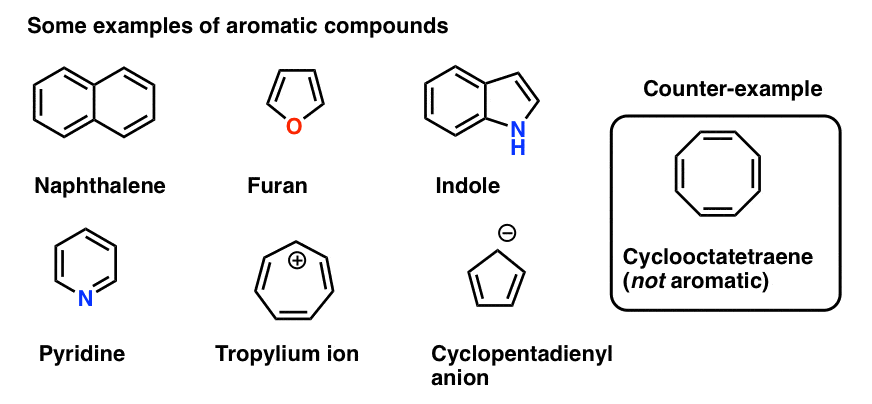
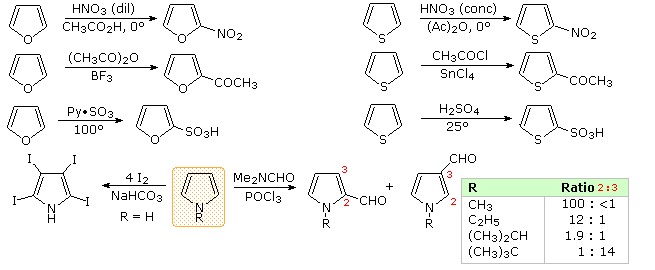
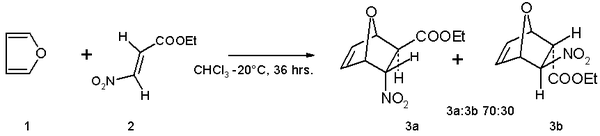
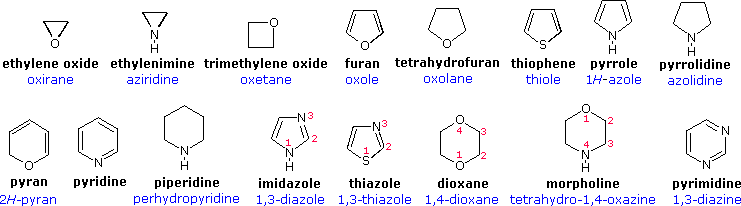
123.Aromaticity(16) – Heterocyclic aromatic systems(2)-Furan,pyrrole and thiophene. – Madoverchemistry

Competition between ring-puckering and ring-opening excited state reactions exemplified on 5H-furan-2-one and derivatives: The Journal of Chemical Physics: Vol 152, No 6

Exploring the Chemistry of Furans: Synthesis of Functionalized Bis(furan‐2‐yl)methanes and 1,6‐Dihydropyridazines - Lopes - 2015 - European Journal of Organic Chemistry - Wiley Online Library

Ring opening reaction of furan with high regio- and diastereo-selectivity via controlled addition of isatin-derived diazoamides - ScienceDirect
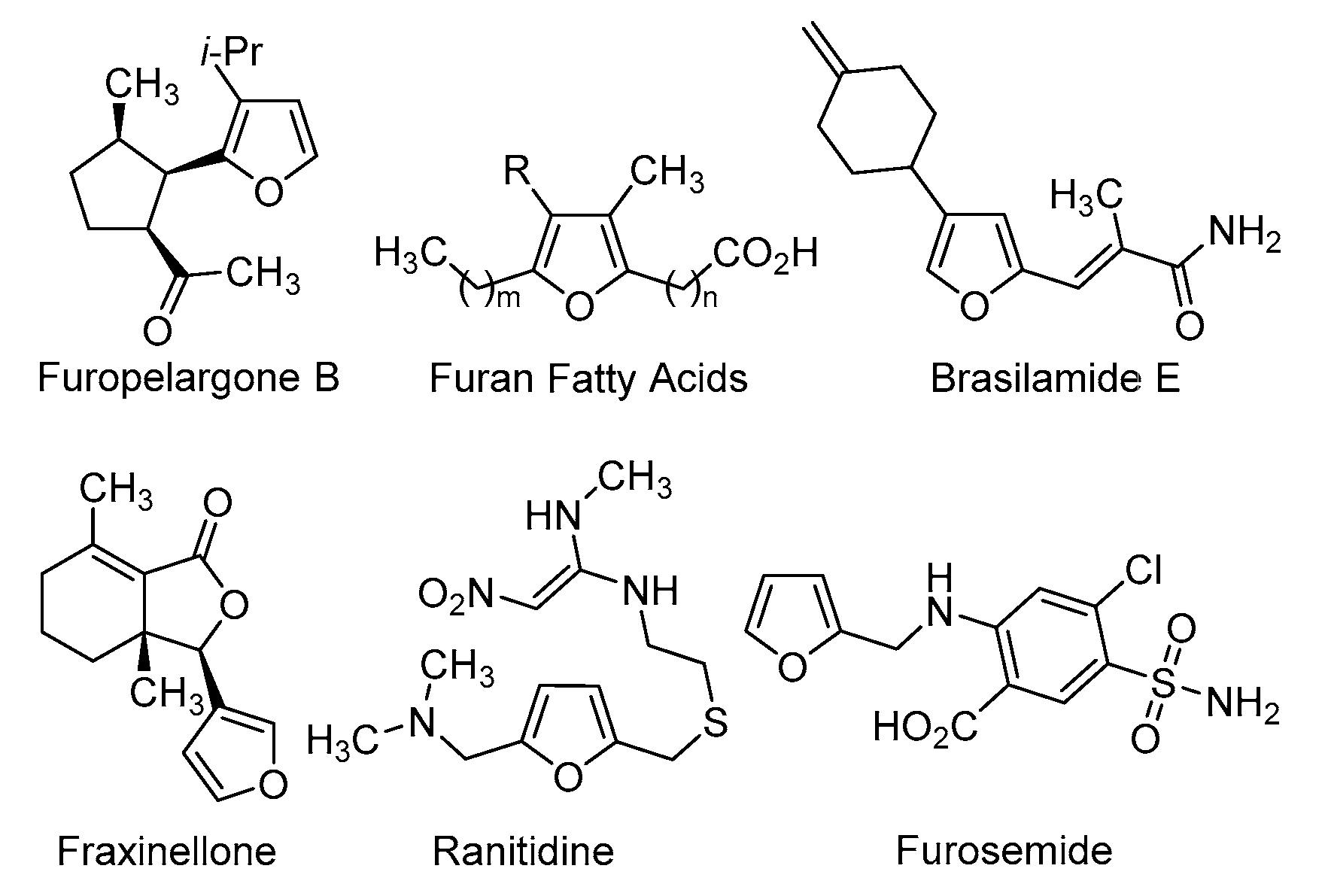
Molecules | Free Full-Text | Transformation of 3-(Furan-2-yl)-1,3-di(het)arylpropan-1-ones to Prop-2-en-1-ones via Oxidative Furan Dearomatization/2-Ene-1,4,7-triones Cyclization | HTML

Theoretical study of photoinduced ring-opening in furan: The Journal of Chemical Physics: Vol 133, No 16
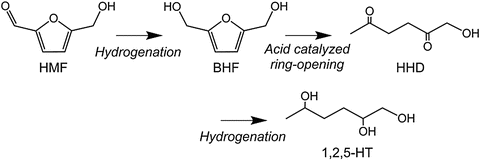
Reductive Conversion of 5-Hydroxymethylfurfural in Aqueous Solutions by Furan Ring Opening and Rearrangement | SpringerLink
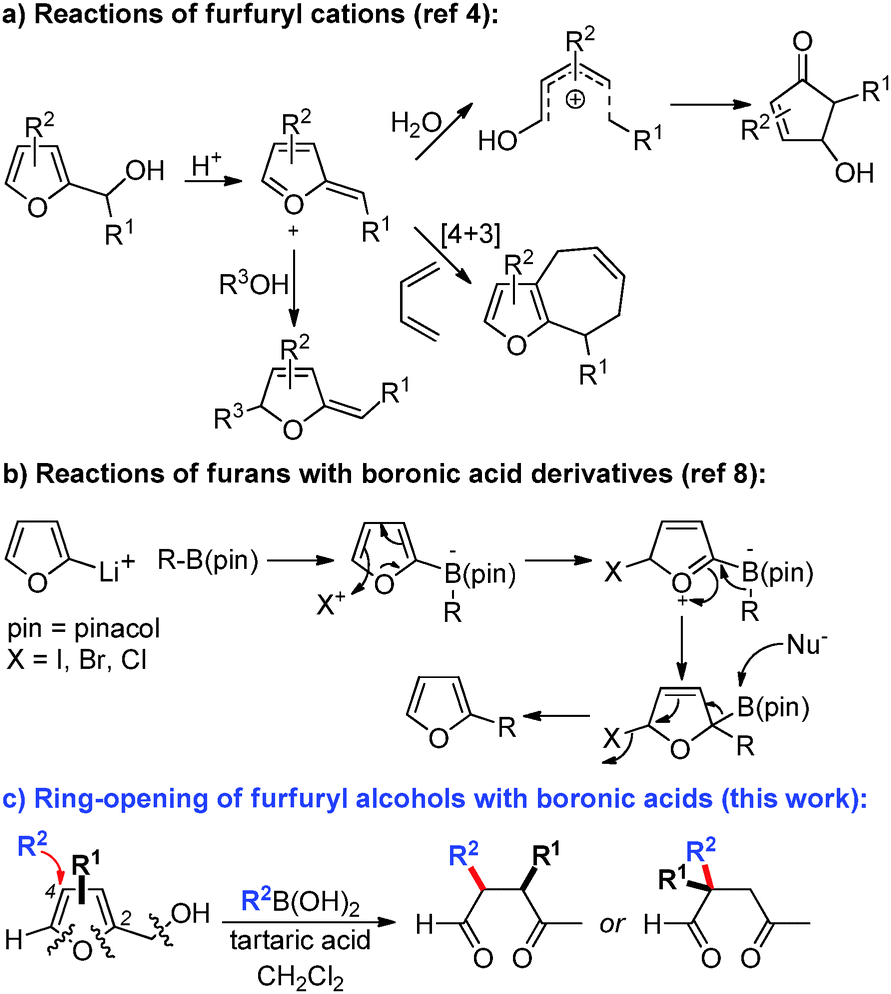
Transition-metal free reactions of boronic acids: cascade addition – ring-opening of furans towards functionalized γ-ketoaldehydes - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC08809G
Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen Stock Photo - Alamy
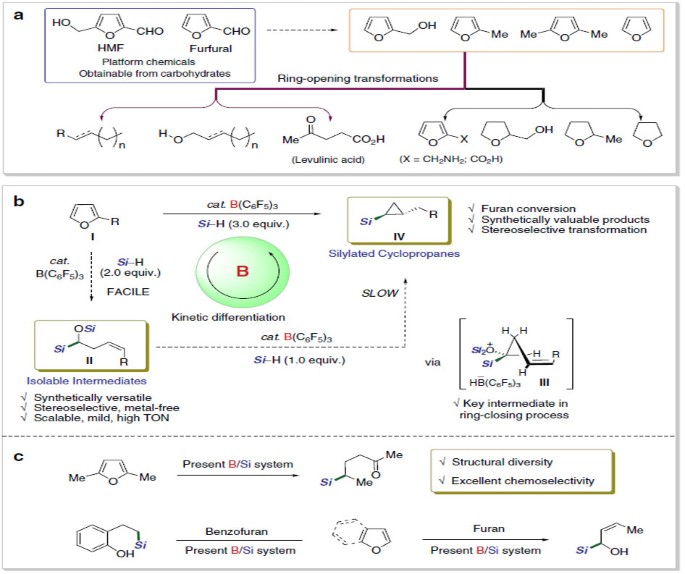

/chapter3/pages41and42/page41and42_files/fivememring.png)