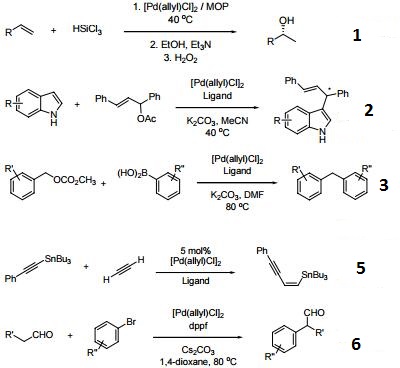
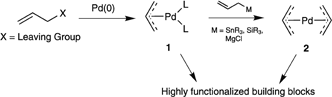
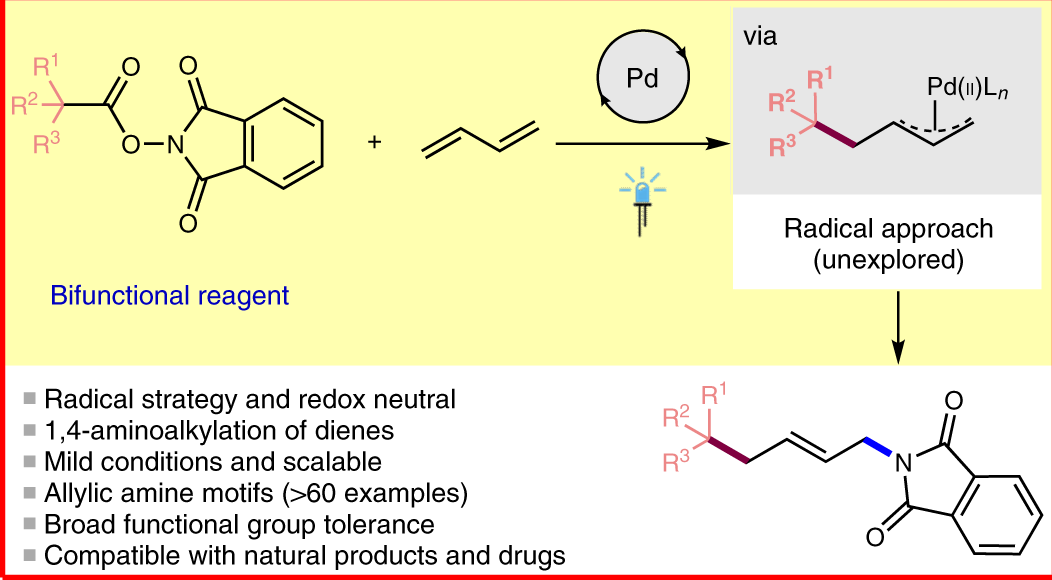
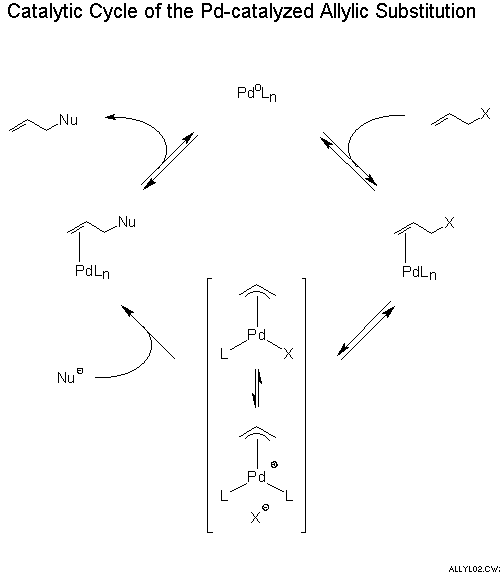
Palladium‐Catalyzed Allylic Substitution at Four‐Membered‐Ring Systems: Formation of η1‐Allyl Complexes and Electrocyclic Ring Opening - Audisio - 2013 - Angewandte Chemie International Edition - Wiley Online Library

π‐Allyl)Pd Complexes Containing N‐Heterocyclic Carbene and Pseudohalogen Ligands – Synthesis, Reactivity toward Organic Isothiocyanates and Isocyanides, and Their Catalytic Activity in Suzuki–Miyaura Cross‐Couplings - Kim - 2013 - European Journal of ...
![PDF] Computational Insights into Palladium-Mediated Allylic Substitution Reactions | Semantic Scholar PDF] Computational Insights into Palladium-Mediated Allylic Substitution Reactions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b392fe8fdff097ec97ca2e5082d62b74ad9d76a8/12-Figure5-1.png)
PDF] Computational Insights into Palladium-Mediated Allylic Substitution Reactions | Semantic Scholar
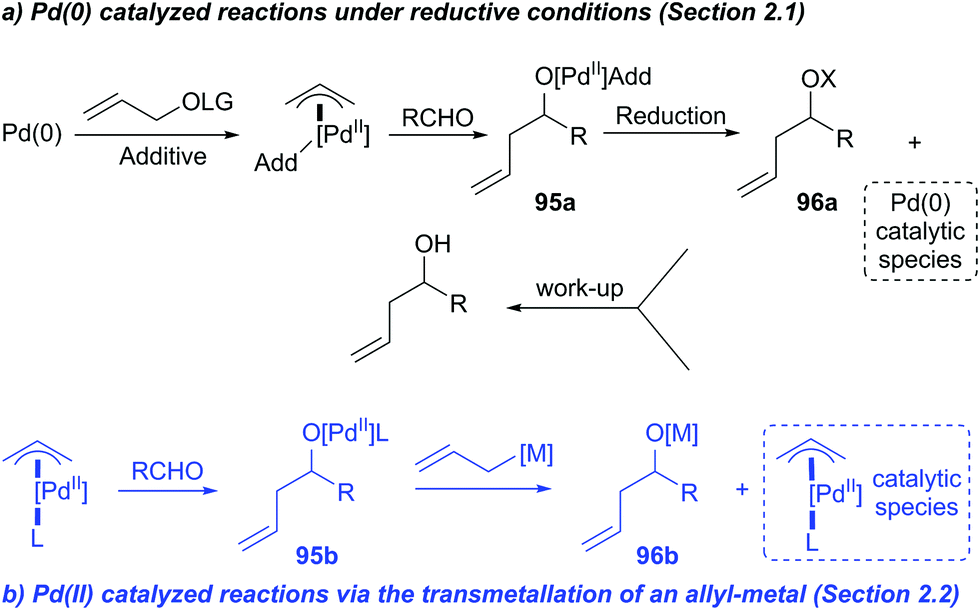
Catalytic nucleophilic 'umpoled' π-allyl reagents - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00449D
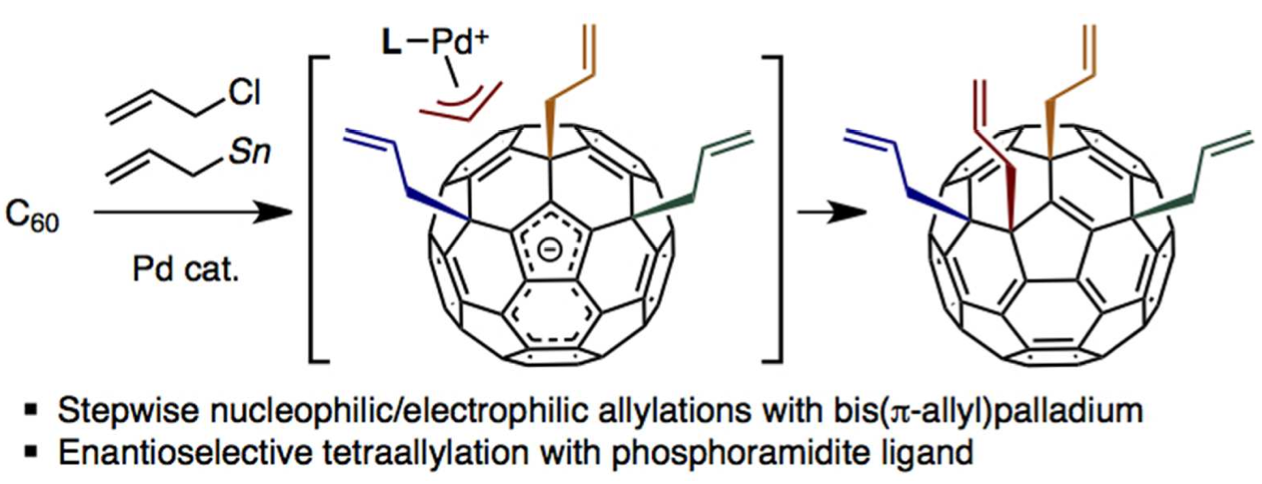
Palladium-catalyzed tetraallylation of C60 with allyl chloride and allylstannane: Mechanism, regioselectivity, and enantioselectivity | Itami Organic Chemistry Laboratory, Nagoya University

Synthesis and characterization of (π-allyl)palladium(II) complexes containing dialkylbiaryl phosphine ligands - ScienceDirect
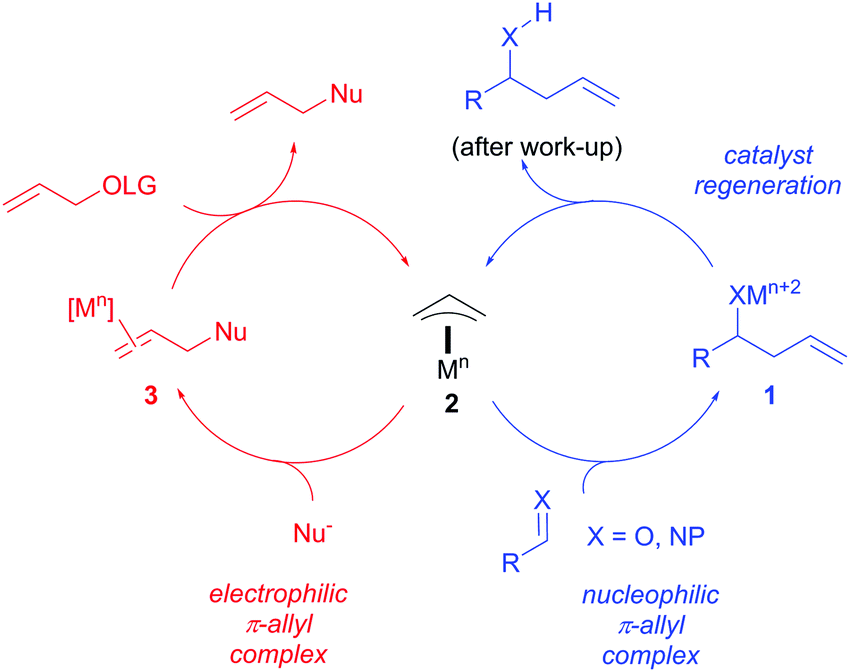
Catalytic nucleophilic 'umpoled' π-allyl reagents - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00449D
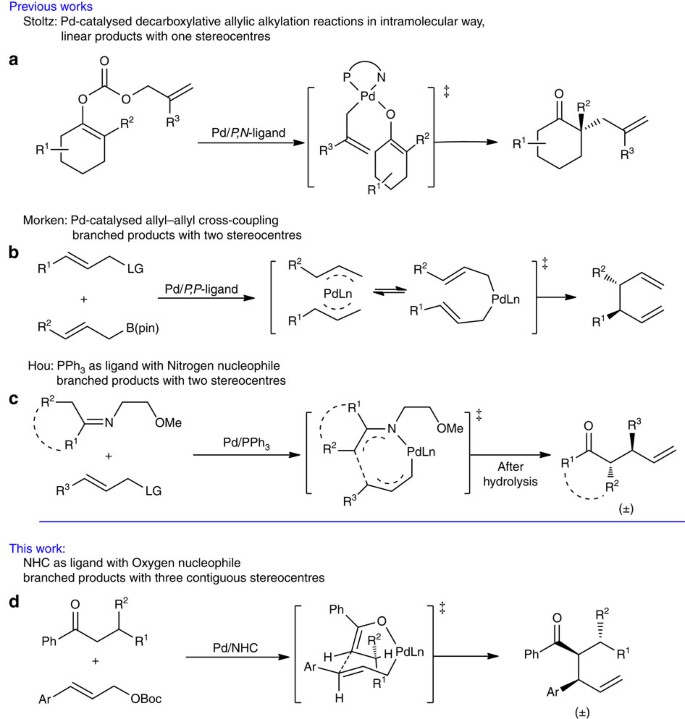
Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications
![Ligand-controlled regiodivergent π-allyl palladium catalysis enables a switch between [3+2] and [3+3] cycloadditions - Chemical Communications (RSC Publishing) Ligand-controlled regiodivergent π-allyl palladium catalysis enables a switch between [3+2] and [3+3] cycloadditions - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C9CC00611G)
Ligand-controlled regiodivergent π-allyl palladium catalysis enables a switch between [3+2] and [3+3] cycloadditions - Chemical Communications (RSC Publishing)

Stereochemistry of the palladium-catalyzed allylic substitution: the syn-anti dichotomy in the formation of (π-allyl)palladium complexes and their equilibration - ScienceDirect
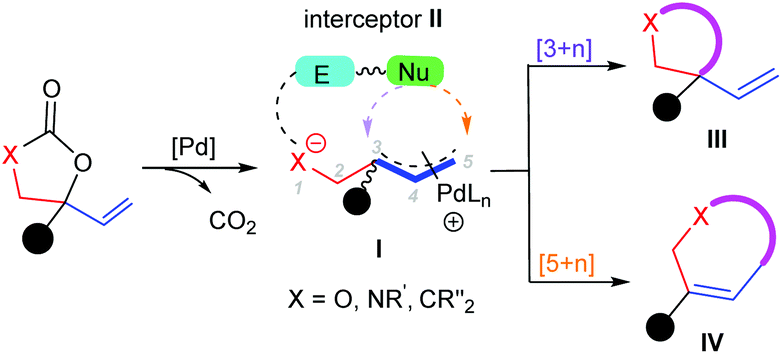
Recent advances in annulation reactions based on zwitterionic π-allyl palladium and propargyl palladium complexes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00273B

Buy Reactivity of Cationic (Pi-Allyl)Palladium(ii) Complexes with Olefins and Dienes. Book Online at Low Prices in India | Reactivity of Cationic (Pi- Allyl)Palladium(ii) Complexes with Olefins and Dienes. Reviews & Ratings - Amazon.in

Mechanism of allyl deprotection through catalytic palladium π-allyl... | Download Scientific Diagram