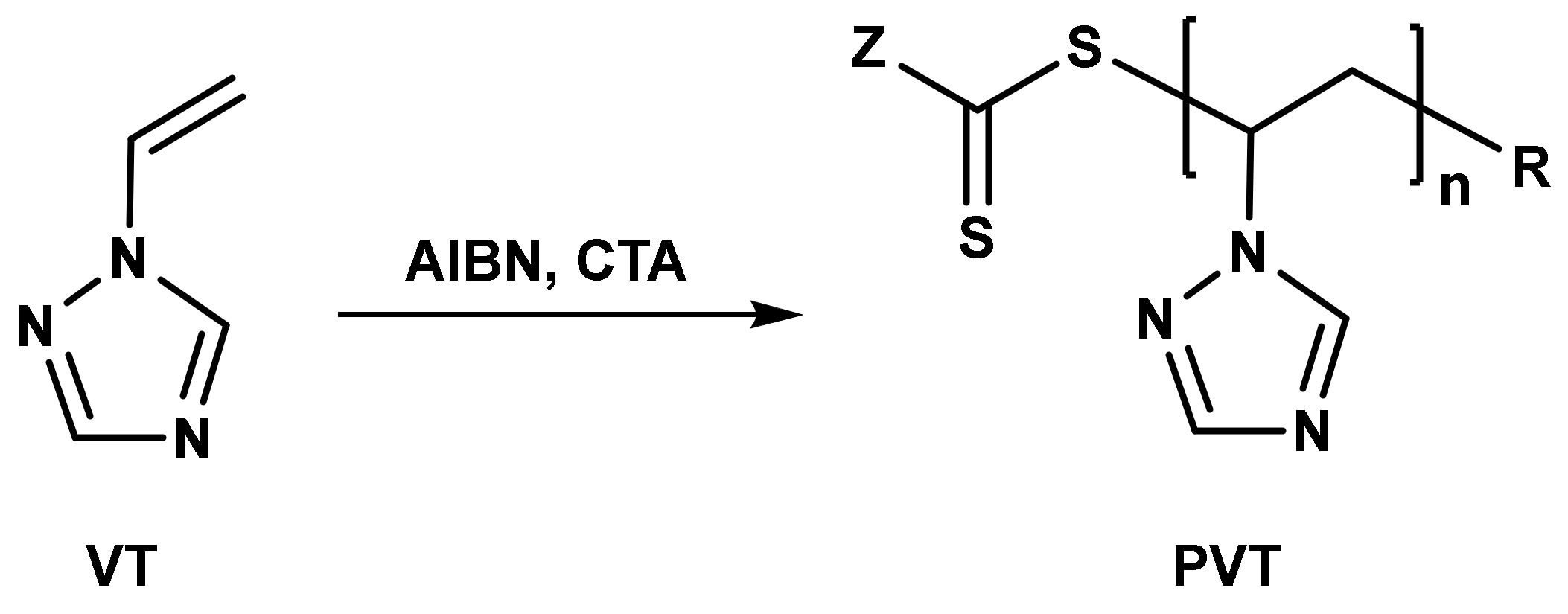
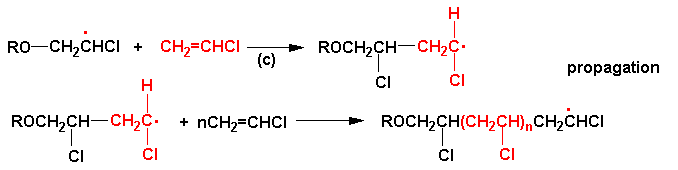
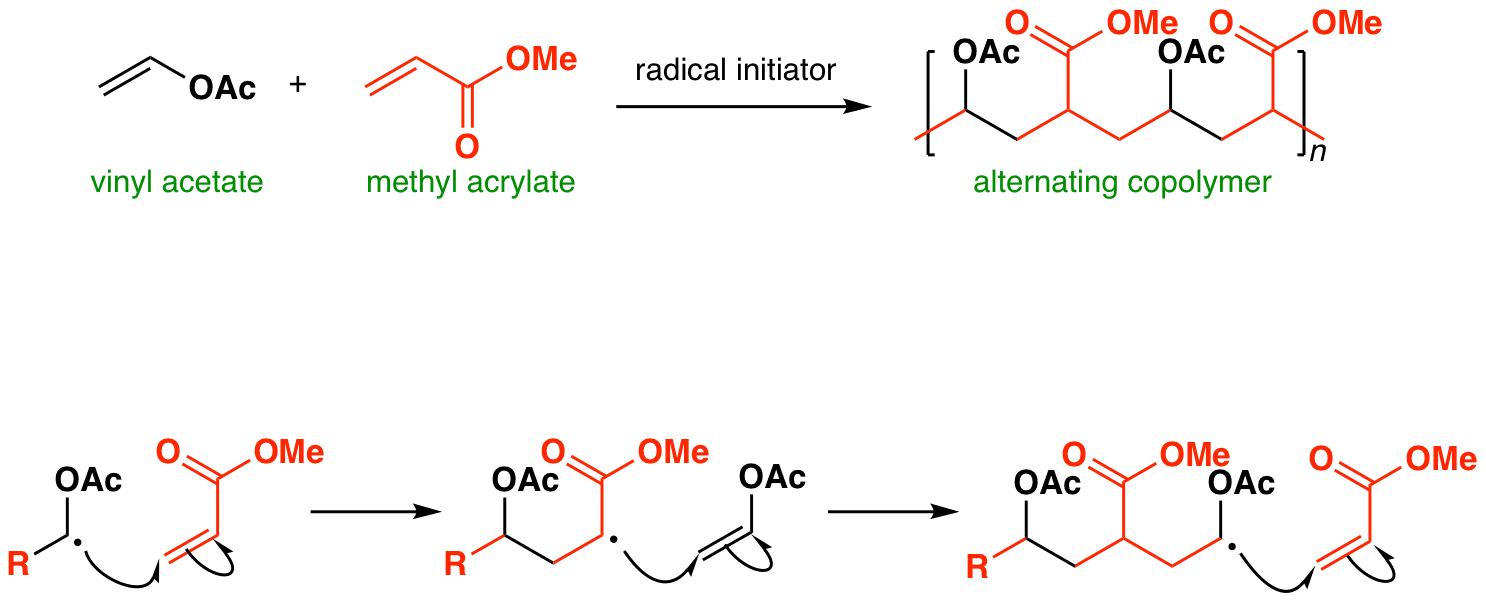
Cascade Vinyl Radical Ipso-Cyclization Reactions and the Formation of α,β-Unsaturated-β-aryl-γ-lactams from N-Propargyl Benzamides | The Journal of Organic Chemistry

Handbook of Radical Vinyl Polymerization (Plastics Engineering): Mishra, Munmaya, Yagci, Yusuf: 9780824794644: Amazon.com: Books
![PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ad4e62b83576af821a221a0a20bd8ad9ee8a9ff9/2-Table1-1.png)
PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar
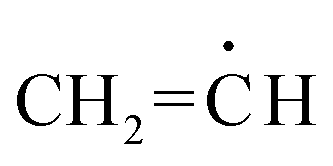
Radicals derived from acetaldehyde and vinyl alcohol - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C7CP04671E

The rate constant for the automerization of vinyl radical: A theoretical approach using the Complete Active Space Self Consistent Field method - ScienceDirect

Copper-Catalyzed Radical Reaction of N-Tosylhydrazones: Stereoselective Synthesis of (E)-Vinyl Sulfones

A remote C–C bond cleavage–enabled skeletal reorganization: Access to medium-/large-sized cyclic alkenes | Science Advances
![PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ad4e62b83576af821a221a0a20bd8ad9ee8a9ff9/1-Figure1-1.png)
PDF] Radical Enamination of Vinyl Azides: Direct Synthesis of N-Unprotected Enamines. | Semantic Scholar

Photochemical Generation and Structure of Vinyl Radicals - Goumans - 2008 - European Journal of Organic Chemistry - Wiley Online Library

Cascade Vinyl Radical Ipso-Cyclization Reactions and the Formation of α,β-Unsaturated-β-aryl-γ-lactams from N-Propargyl Benzamides | The Journal of Organic Chemistry

Cascade Vinyl Radical Ipso-Cyclization Reactions and the Formation of α,β-Unsaturated-β-aryl-γ-lactams from N-Propargyl Benzamides | The Journal of Organic Chemistry
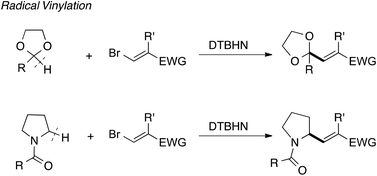
Radical vinylation of dioxolanes and N-acylpyrrolidines using vinyl bromides - Organic Chemistry Frontiers (RSC Publishing)

Photochemical Generation and Structure of Vinyl Radicals - Goumans - 2008 - European Journal of Organic Chemistry - Wiley Online Library
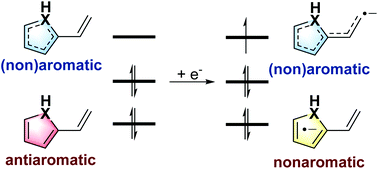
Antiaromaticity-promoted radical anion stability in α-vinyl heterocyclics - Organic Chemistry Frontiers (RSC Publishing)